HOWARTH LAB

SOCIAL
ADDRESS
Department of Pharmacology,
University of Cambridge,
Tennis Court Road,
Cambridge
CB2 1PD
UK
CONTACT
e: mh2186@cam.ac.uk
t: 01223 334176
2026 Mark Howarth. All rights reserved.
The theme of our research is Innovating Protein Technologies for Therapeutics and Vaccine Design. We have a range of project areas
running in the lab, from fundamental understanding of protein interactions and reactivity through to clinical application. The lab is also
enthusiastic about entrepreneurship, supporting team members to create new concepts and develop the potential of their new
technologies to tackle important challenges.
Immuno-engineering and Global Health
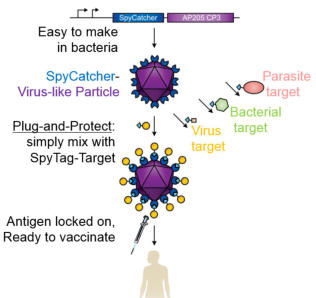
Developing an effective vaccine may be the most effective way to improve human health. The group has established a route to
accelerate vaccine development, through our Plug-and-Protect platform. A limiting factor in vaccine generation is the difficulty of
turning a promising target protein into the kind of assembly that would give long-lasting disease protection. We showed rapid and
efficient decoration of virus-like particles, which elicited a strong immune response even with only a single injection. We demonstrated
potent immunization towards the global health challenge of malaria, working with collaborators at Oxford University’s Jenner Institute.
This approach is now being used by many groups against cancer and diverse infectious diseases, e.g. HIV, influenza, tuberculosis, and
veterinary pathogens. Tag/Catcher vaccine technology has progressed to Phase 3 clinical trials for Covid-19, with clinical trials under
way against malaria and CMV (a major cause of deafness and blindness in babies). Our current focus in this area is to create new
protein antigen and nanoparticle strategies, to achieve broadly protective immune responses against targets that evade regular
immune responses. We are working to establish new routes to general mucosal immunity, as well as to protect from gastric cancer. We
have ongoing collaborations with Oxford, Caltech and the NIH, working towards protection against the most urgent global health
challenges.
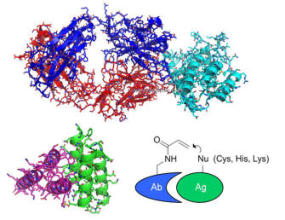
Synthetic Biology and Click Biology from a New Generation of Protein Interactions
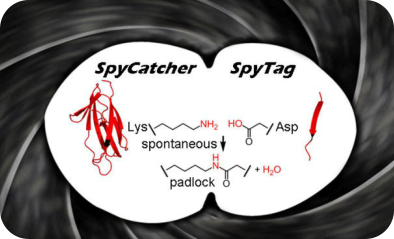
We have harnessed an amazing feature of the surface of the pathogenic bacterium Streptococcus pyogenes, to create a family of protein
superglues. This natural protein reactivity enabled us to form a spontaneous isopeptide bond between genetically-encoded protein
and peptide partners. Our favourite pair, SpyTag/SpyCatcher, is one of the strongest protein interactions ever measured. SpyTag is now
applied by more than a thousand labs around the world for diverse areas of basic research and biotechnology. The group also
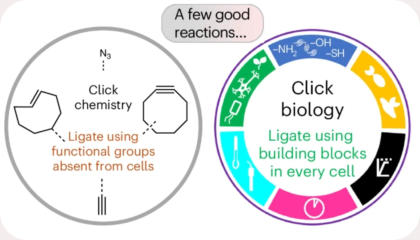
introduced the concept of Click Biology, to describe how easy-to-use genetically-encoded reactivity may empower a revolution in
biological research, as happened in the physical sciences through Click Chemistry. We have now developed interactions tunable by pH
and light. Combining computational design and evolution through phage display, we created the first genetically-encoded interaction
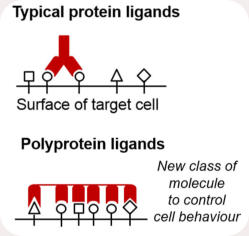
reacting at the diffusion limit and approaching infinite affinity. We are extending this new class of protein interaction, to create unique
new possibilities for synthetic biology. We are building a rainbow of protein superglues, to pattern interactions within cells and
between cell-types. SpyTag provides unique opportunities for building resilient enzyme teams, for green biotransformation. Click
Biology is a powerful tool to overcome challenges in gene therapy targeting and cancer evasion of cell therapy. SpyTag accelerates the
creation of antibody teams for combinatorial control of cell signalling, for more potent targeting of cancer. We are now integrating
protein binder assemblies with successful small molecule drugs, to achieve a new level of precision in control of cell fate and unveil
new therapeutic possibilities.
Vaccine development:
Unique Protein Architectures and Chemical Biology for Cell Therapy
Our studies defining the limits of cancer cell capture from blood made clear that even the best antibody interactions are not good
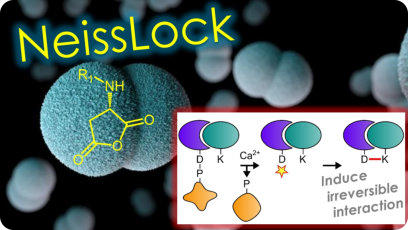
enough. We have developed a new class of binding proteins that form covalent bonds to endogenous protein targets. NeissLock was
engineered from an adhesion system from Neisseria meningitidis and forms an anhydride in response to calcium, reacting irreversibly
with neighbouring proteins. Protein ligands that never let go of their targets should reduce the detection limit of soluble biomarkers
for early diagnosis. We are progressing NeissLock technology to generate long-acting therapeutics through hitchhiking on red blood
cells. We are also working to enhance CAR-T cell therapy of cancer, which has been revolutionary against leukaemia/lymphoma but has
had little success against solid tumours.
Get in contact for further information about any of these projects, or to discuss the possibility of working on other projects in the area
of synthetic biology / vaccines / chemical biology / cancer.